Definition:
- A refinery is a facility designed to convert crude oil into refined products.
Quick Facts:
- The first refinery in the United States was built in 1876, near Pico Canyon, CA. The refinery boiled oil to produce kerosene, lubricants, and waxes. Gasoline was only produced as a by-product at that first refinery, and was discarded.
- With the introduction of the gasoline-powered automobile in 1892, a demand for gasoline began to develop.
- In 2003, refineries in the United States processed an average 15.3 million barrels of crude oil per day, from which 8.5 million barrels of gasoline was produced.
- Other by-products of the refining process are jet fuel, kerosene, propane, diesel fuel, and home heating oil.
- Refinery products are also used to make plastics, fertilizers, synthetic fibers, paint, and other everyday products.

How does a refinery work?
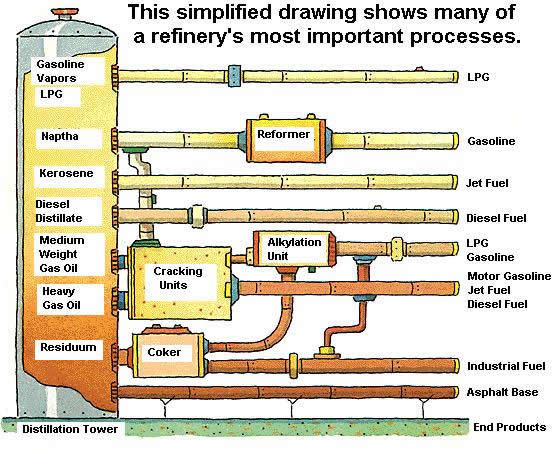
While refinery processes have advanced since the time of Pico Canyon, the basic process still remains the same. The process of converting crude oil into useable by-products involves three stages: separation, conversion and treatment.
Separation

Separation begins with vaporizing crude oil, by pumping it through pipes in hot furnaces.
The vapors separate inside the towers, based on density and boiling point. Light vapors rise to the top of the tower and the heavier vapors descend to the bottom. The vapors then condense back into liquids. Gasoline, being one of the lighter products produced by the refinery, layers at the top of the distillation tower; diesel fuel and kerosene in the middle, and heavier products like gas oils and asphalt products accumulate at the bottom.
Conversion
Conversion is the process of transforming the output of distillation towers into streams that eventually become finished products. Techniques of conversion are illustrated below. Some products emerge from the distillation process as ready-to-use products such as gasoline, and liquefied petroleum products. Others require further conversion.
One method of conversion is called cracking. Heat and pressure are applied in the cracking unit to break heavy hydrocarbon molecules into lighter molecules. In the diagram below, cracking is used to convert gas oils into motor gasoline, jet fuel, and diesel fuel.
In the alkylation unit, a catalyst is added under high temperature and low pressure to crack heavy molecules into lighter gasoline and liquefied petroleum gas molecules. A catalyst is something that is added to the process to achieve a chemical reaction but is not consumed by the reaction.
In a coker, heat and moderate pressure is used to convert process residues into industrial fuel, and products that can be further refined through other conversion processes such as alkylation.
Treatment
To meet government regulations, many products must go through further processing to remove impurities, such as sulfur and nitrogen, and to meet federal and state air pollution requirements. The emission of carbon monoxide is reduced through oxygenation. Oxygenation is achieved through the addition of ethanol, or other additives, to gasoline.
Gasoline may also be treated to boost octane ratings. An octane rating is a measurement of how well a gasoline will perform in an automobile engine. Much of the gasoline that comes from cracking units does not have a high enough octane rating to perform in today’s automotive engines.
The low-octane gasoline process streams are sent to reforming units where their octane levels are boosted. Using catalytic converters, hydrocarbon molecules are “reformed” into high octane gasoline components.
Date of Revision: 12012011
